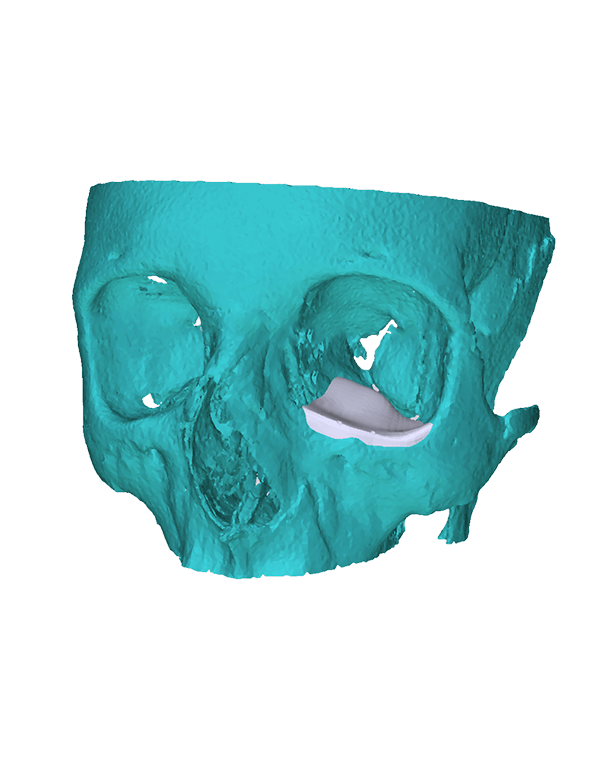
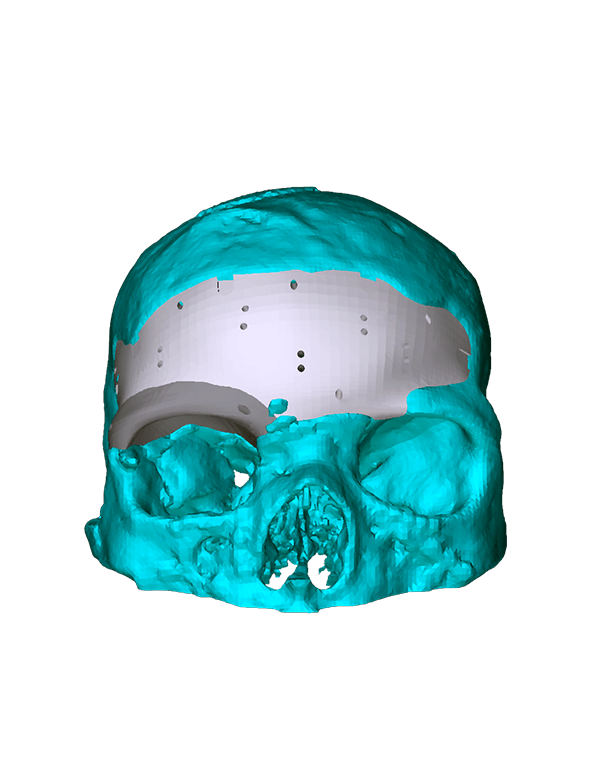
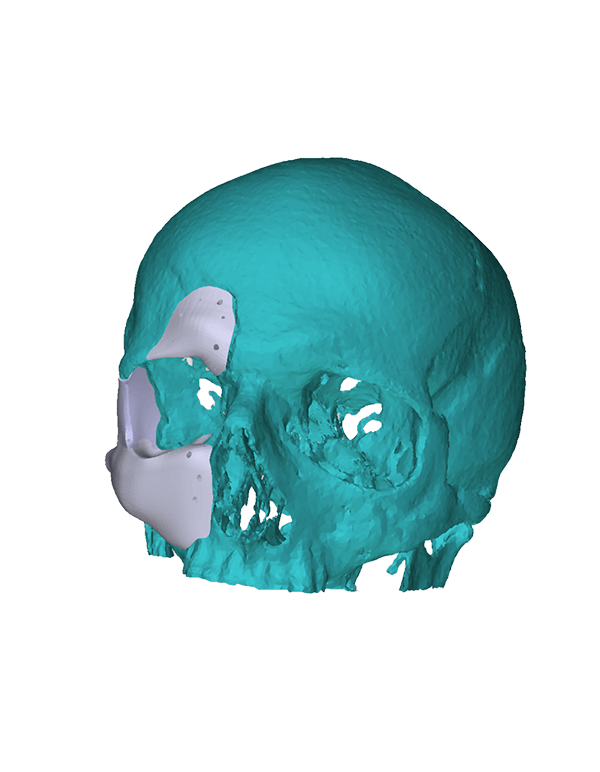
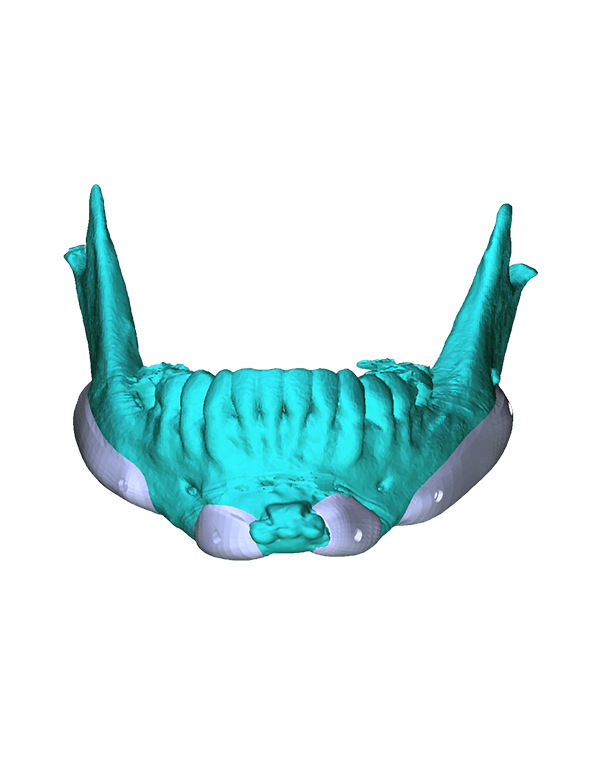
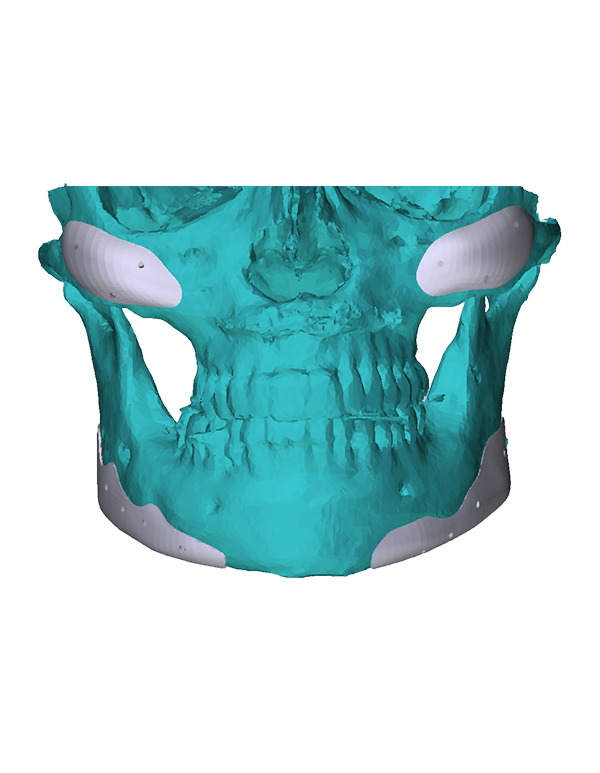
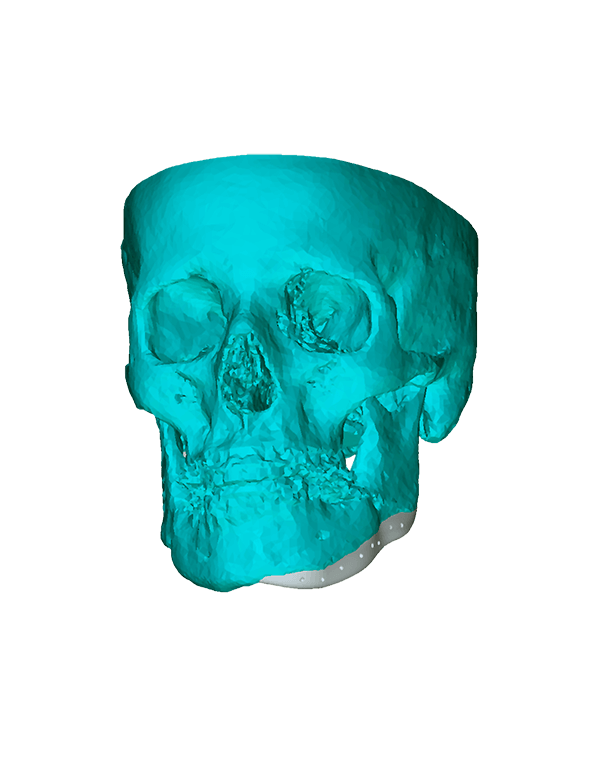
Patient-specific
implants
All about
3di cranioplastics
3di GmbH has developed its own process for manufacturing individual CAD implants. Taking into account the surgical aspects, it is possible to manufacture patient-specific implants within a very short period of time. The main areas of application for our implants are: neurosurgery, trauma surgery, tumour surgery, reconstructive and aesthetic plastic surgery, oral and maxillofacial surgery, and ear, nose and throat medicine.

Implant material used
The material is selected based on medical and manufacturing considerations. Only medically approved materials are used.
BIOVERIT®II
is a dense, non-outgassing and non-resorbable material that can be machined with conventional tools with high manufacturing accuracy. BIOVERIT®II is a machinable glass ceramic of the SiO2-Al2O3-MgO-Na2O‑K2O‑F system, consisting of a crystal phase (approx. 60 %) with mainly phlogopite crystals embedded in an aluminosilicate glass matrix (approx. 40 %). No toxic effects are known.
PEEK
Polyetheretherketone was developed as a non-resorbable high-performance thermoplastic for long-term implantation and is an aromatic polymer with a linear structure belonging to the (poly)aryletherketones (polymer group). PEEK is a semi-crystalline material that can be machined with high manufacturing accuracy using conventional tools. Since 2009, 3di GmbH has been producing patient-specific implants for the skull from this material.
TITANIUM
is a non-resorbable, highly stable metal for long-term implantation and has already proven itself in many areas of the body. Due to this stability, it is possible to design the implants to be very thin. Subsequent machining with surgical tools is very difficult or even impossible due to its high strength. Since pure titanium tends to wear down and is bendable, we use the titanium alloy Ti-6Al-4V for our implants.
Our procedure
Data creation and data transfer
The necessary data of the defect is recorded in the medical facility using computer tomography imaging and then stored in DICOM format.
The data is sent to us securely, encrypted and quickly via our internet transfer portal. You can also provide the data as a QR code by email or send it to us by post on a data carrier (CD-ROM, DVD, USB stick).
Please enquire here for the desired CT scan parameters and the link to the internet transfer portal.

Data preparation
We process and convert the data from the patient’s CT scan to create a virtual 3D model of the patient’s head.

Creation of a 3D implant model
An implant model is constructed on the basis of the virtual 3D patient model. After consultation between the attending physician and our design engineers, you [the physician] determine the final implant shape. If required, we will also send you physical 3D models for implant planning.

Sending approval and commercial order to 3di
In order to produce the cranioplasty, we require two documents in every case. The approval of the design (“agreement protocol”) by the attending physician, as well as a commercial order specifying the delivery and billing address.
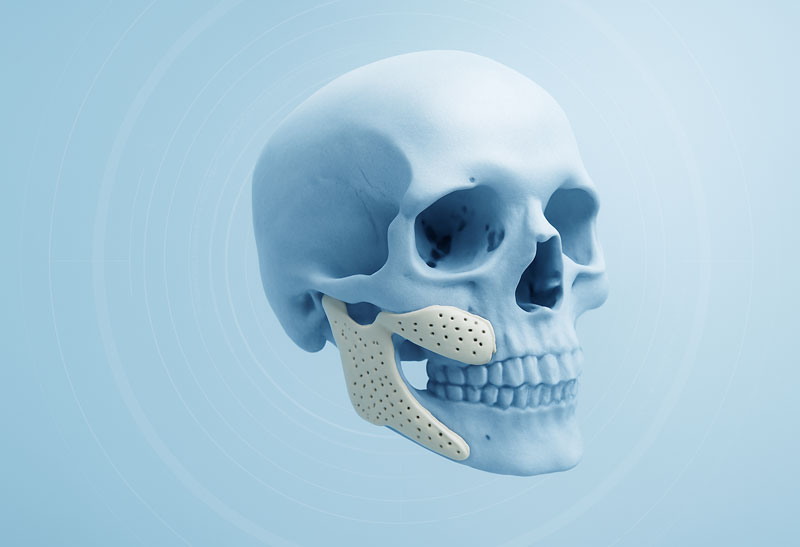
Manufacture and dispatch of the implant
Once the implant shape has been approved, the design data is converted into CNC control data and then manufactured using computer-aided production in our in-house production centre.
Finally, the implant is shipped together with the necessary paperwork and any necessary accessories, such as resection templates or special screws for fixation.

Frequently asked questions
How complex can the implants be?
Almost any bone structure of the human skull can be replicated. The manufacturing accuracy is < 0.5 mm. The quality of the CT data provided to us (age, layer spacing) is decisive.
What aspects are taken into account when designing the shape?
Functional protection is ensured and the patient’s external shape is restored or significantly improved in the case of genetic defects.
Can soft tissue deficits be compensated for with the implant/augment?
If the patient’s skin condition allows it, soft tissue deficits can be compensated for if necessary. However, this should definitely be discussed in advance with the responsible designer.
How can the implants be sterilised?
As the implants are delivered non sterile, detailed cleaning and sterilisation instructions (steam sterilisation, 134 °C) are included with every CAD plastic sent. Our implants are suitable for re-sterilisation (max. 2 reprocessing cycles). However, the decision on whether to reuse them on a patient is always the responsibility of the treating physician.